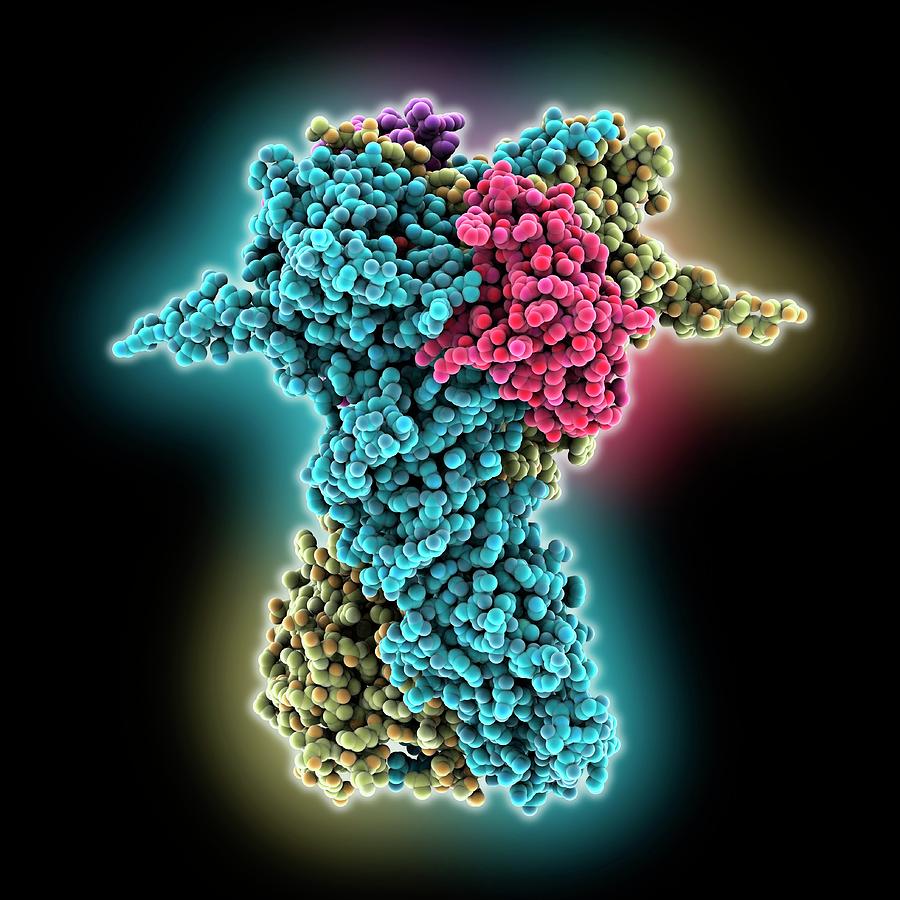
Understanding these reactions is of great interest in the light of the many neurodegenerative diseases in which folding goes awry. It is expressed in a wide variety of organisms from bacteria to humans. The message in this RNA is translated into strings of amino acids proteins. What are Chaperones DNA is a linear chain of nucleotides, portions of which are faithfully transcribed into linear messenger RNA. Similar but even more complicated chaperone networks control the proteome of human cells. In molecular biology, chaperone DnaJ, also known as Hsp40 ( heat shock protein 40 kD), is a molecular chaperone protein. Chaperones are a functionally related group of proteins assisting protein folding in the cell under physiological and stress conditions. DnaK is also involved in chromosomal DNA replication, possibly through an analogous interaction with the DnaA protein. These subunits form a cage-like structure. “Apparently, DnaK realises that the attached protein chains will never be able to mature into useful molecules,” says the biochemist. Plays an essential role in the initiation of phage lambda DNA replication, where it acts in an ATP-dependent fashion with the DnaJ protein to release lambda O and P proteins from the preprimosomal complex. What is a chaperone protein and what is its function Chaperonin proteins are types of Chaperone proteins which contain two main subunits, Hsp60 and Hsp10. For example, when GroEL is removed from the cells, its client proteins accumulate on DnaK, which then shuttles them to proteases to be decomposed. Chaperones are a functionally related group of proteins assisting protein folding in the cell under physiological and stress conditions. The researchers also investigated what happens when the chaperone network is disturbed. It forms a nano-cage in which a single protein chain is temporarily enclosed and allowed to fold while protected from external influences. GroEL is a highly specialised folding machine. Those it cannot fold are transferred to yet another chaperone, the barrel-shaped GroEL. Furthermore, DnaK mediates the folding of most of these protein chains. “It functions as a kind of turntable.” DnaK binds to about 700 different protein chains as they are synthesised. “We identified the Hsp70 protein DnaK as the central player of the network,” explains Ulrich Hartl. Increased chaperone expression can suppress the neurotoxicity caused by protein misfolding, suggesting that chaperones could be used as possible therapeutic agents. Using proteomic analyses they show how different chaperones cooperate during the folding process. MPIB scientists have now investigated the organisation of this network in the bacterium Escherichia coli. All cells, from bacteria to human, have therefore developed a network of molecular chaperones, proteins themselves, which help other proteins to fold properly. Cryo-electron microscopy structure of the soluble membrane attack complex shows how chaperone protein, clusterin (pink), helps to prevent immune activation complexes (blue) from damaging human cells. 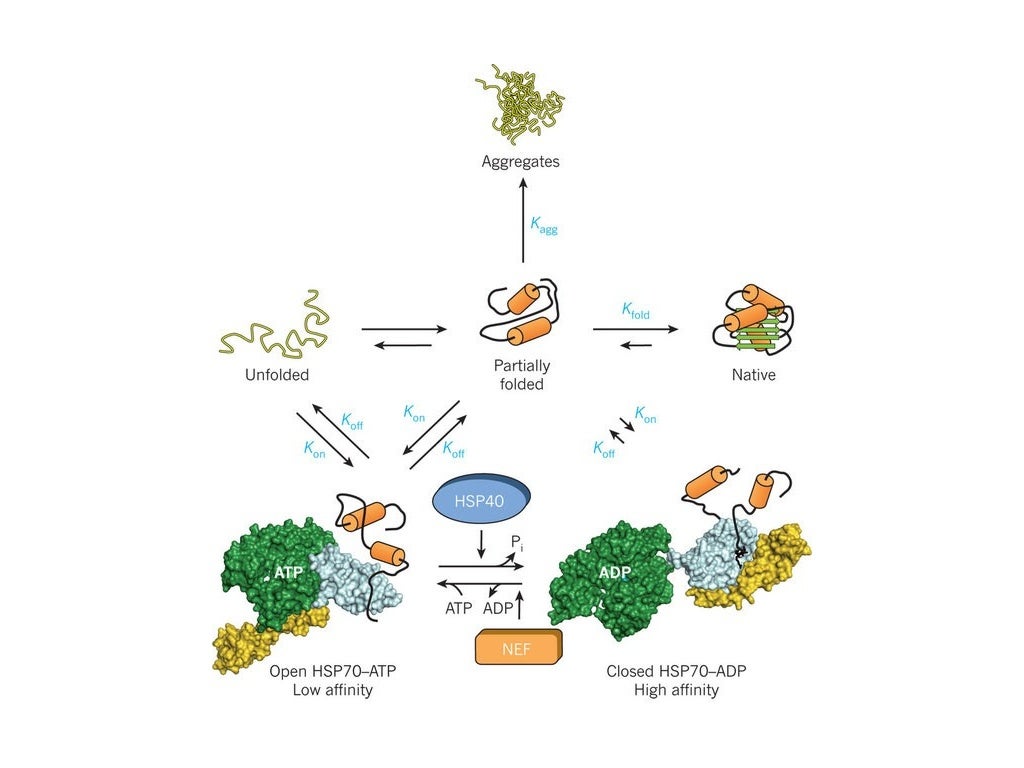
Hsp70 is one of the bodys many chaperone proteins, which promote the proper folding. When this difficult process goes wrong, it can lead to useless or even dangerous protein clumps. Presence of protein extends life potential aging mechanism found. In order to be biologically useful, these chains must fold into a complex three-dimensional pattern. The cells of the human body continuously synthesise thousands of different proteins in the form of amino acid chains. This can naturally happen with chaperones as well.Proteins are responsible for almost all biological functions. 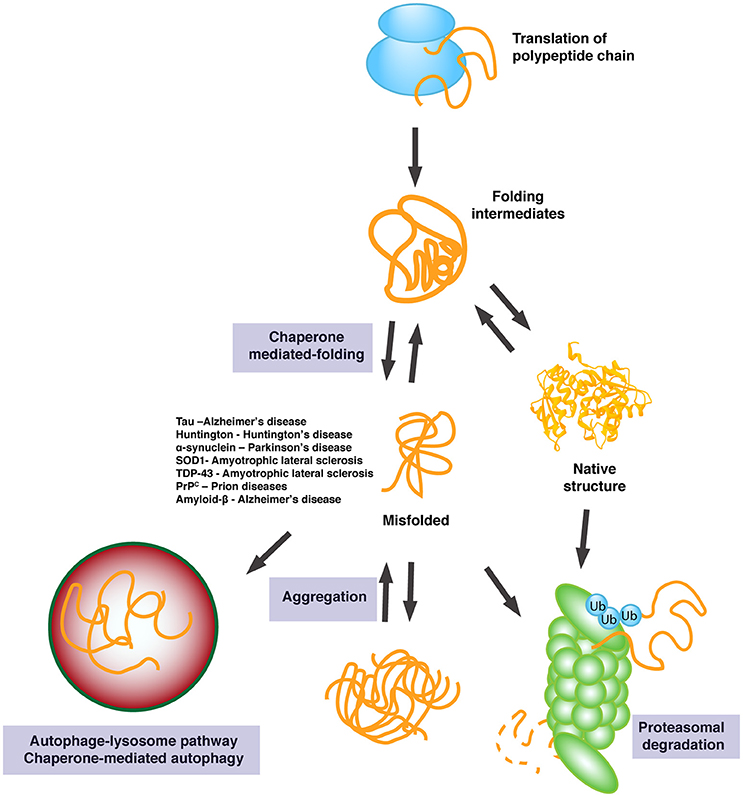
This can happen with a transcription error during mRNA synthesis since each mRNA molecule is read by many ribosomes. * It's not uncommon for a "bad" batch of proteins to be created. Not all chaperones are resistant to heat though and many are no more intrinsically resistant to denaturing. This is true because they need to be able to withstand noxious conditions that denature other proteins. Some chaperones, especially heat-shock proteins, may be more resistant to misfolding. After protein denaturation caused by stress (for example, due to heat or toxin exposure) or disease conditions, proteins can be unfolded, disaggregated and then refolded, or they can be targeted for disposal by proteolytic systems. If this is happening too often and the number of chaperones drop too low or the number of unfolded or incorrectly folded proteins becomes excessive *, the unfolded protein response may be triggered and, if it does not resolve the issue and the cell remains stressed, the cell will undergo apoptosis and die. A set of protein families termed molecular chaperones assists various processes involving folding, unfolding and homeostasis of cellular proteins. If that happens, it will either be assisted by another chaperone and given time to fold successfully or it will be destroyed. Chaperone proteins are still proteins and they can certainly misfold just like any other. Molecular chaperones seem to be especially important for the maintenance of protein complexes and pathways that are closely associated with nuclear activities.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
